 [Wyomissing, PA] – June 17, 2015 –Francisco L. Tellez, MD, FACS and Domenic C. Izzo, Jr., MD of Berks Eye Physicians & Surgeons. Ltd. announced that they are providing the iStent® Trabecular Micro-Bypass implant as a treatment option to reduce eye pressure for patients with both cataracts and glaucoma.
[Wyomissing, PA] – June 17, 2015 –Francisco L. Tellez, MD, FACS and Domenic C. Izzo, Jr., MD of Berks Eye Physicians & Surgeons. Ltd. announced that they are providing the iStent® Trabecular Micro-Bypass implant as a treatment option to reduce eye pressure for patients with both cataracts and glaucoma.
The iStent® is FDA-approved for use in conjunction with cataract surgery to reduce eye pressure in adult patients with mild-to-moderate open-angle glaucoma currently being treated with glaucoma medicine. In clinical trials, iStent® has been shown to safely reduce eye pressure; elevated eye pressure is the main cause of open-angle glaucoma.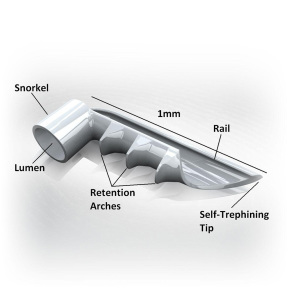
“This is great news for people who have both a cataract and glaucoma,” says Dr. Tellez. “The drops used to treat glaucoma can be difficult to administer and expensive. With iStent®, many patients will need less medicine to control their eye pressure and some may not need prescription eye drops at all.”
iStent® is the smallest medical device ever approved by the FDA and is implanted in the patient’s eye during cataract surgery. Dr. Izzo added, “Since it is so small and is placed inside the eye you are unable to see or feel it after the procedure is done. Although you won’t even know the iStent® is there, it will be working to help reduce your eye pressure.” Implanting the iStent® does not significantly extend the length of time the patient spends in surgery and has a safety profile similar to cataract surgery alone.
 Because of cost, side effects, or even forgetfulness, a high percentage of glaucoma patients do not use their medicines properly, and many discontinue them altogether. Dr. Tellez, fellowship-trained glaucoma specialist, cautions that “This can be a serious problem. When pressure in the eye is out of control, it can increase the risk for permanent vision loss. I consider iStent® to be an important advancement in protecting against vision loss for my glaucoma patients.”
Because of cost, side effects, or even forgetfulness, a high percentage of glaucoma patients do not use their medicines properly, and many discontinue them altogether. Dr. Tellez, fellowship-trained glaucoma specialist, cautions that “This can be a serious problem. When pressure in the eye is out of control, it can increase the risk for permanent vision loss. I consider iStent® to be an important advancement in protecting against vision loss for my glaucoma patients.”
 1802 Paper Mill Road, Wyomissing, PA 19610
1802 Paper Mill Road, Wyomissing, PA 19610

